On February 7, a team led by Professor Dong Bo from the Fang Zongxi Center for Marine Evo-Devo at Ocean University of China (OUC) published a research article entitled “Identification of protein precursor for thyroid hormone synthesis in basal chordate ascidian Styela clava” in the internationally renowned journal Nature Communications. The study is the first to identify and functionally validate, in an invertebrate, the protein precursor required for thyroid hormone biosynthesis, thyroglobulin-like (TG-like), thereby providing important scientific evidence for understanding the origin and evolution of the thyroid hormone endocrine system.
Thyroid hormones (THs) play central regulatory roles in animal growth and development, metabolic regulation, and life-cycle transitions. The mechanism of TH synthesis is systematically elucidated in vertebrates with thyroglobulin (TG) as the protein precursor for hormone synthesis. TG provides the key tyrosine residues required for TH production and serves as a core molecule in this process. However, although THs have been detected in a variety of invertebrates and their important roles in development and metamorphosis have been validated, the key protein precursor involved in TH synthesis has remained unidentified. This long-standing gap has constrained our understanding of the molecular basis of TH synthesis and the evolutionary origin of the thyroid endocrine system.
Using the chordate Styela clava as the research subject, the researchers combined immunofluorescence, immunoprecipitation-mass spectrometry, phylogenetic analysis, expression analysis, and in vitro iodination experiments to successfully identify the protein precursor for TH synthesis in this ascidian, which they named thyroglobulin-like (TG-like). The study found that the TG-like protein contains multiple Tg1 domains (vertebrate TG proteins contain 11 Tg1 domains). Compared with other Tg1 domain-containing proteins, TG-like is more closely related to vertebrate TG proteins. Expression analyses further showed that the spatiotemporal expression patterns of the Tg-like gene during early development of S. clava were highly consistent with those of key genes involved in TH synthesis, and it was specifically expressed in the thyroid-equivalent region of the endostyle, the structure responsible for TH synthesis in ascidian adults. In vitro iodination experiments further demonstrated that the TG-like protein is capable of providing hormonogenic sites for TH synthesis.
The research team conducted in vivo RNA interference (RNAi) experiments to knockdown the expression of Tg-like gene in S. clava. It was observed that TH (T3 and T4) levels were decreased significantly upon knockdown of the Tg-like gene, and that key developmental events during metamorphosis, including tail regression, organ morphogenesis, and body extension, were all remarkedly impaired. It is noteworthy that exogenous supplementation with TH effectively rescued the developmental defects and abnormal phenotypes caused by Tg-like knockdown. These results indicate that the Tg-like gene plays a key regulatory role in the metamorphosis of S. clava by participating in the synthesis of TH.
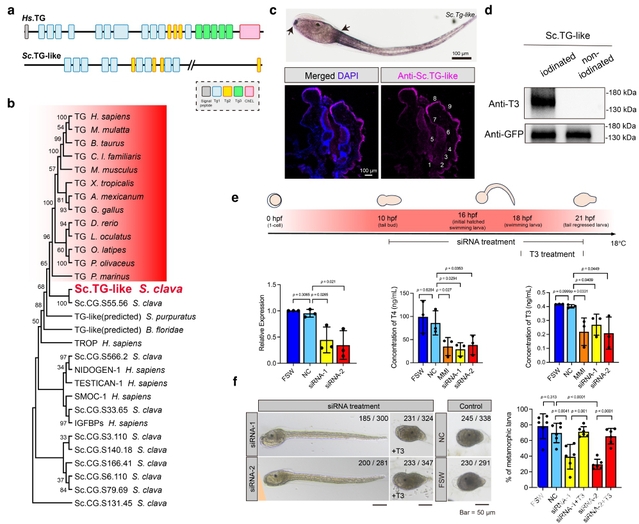
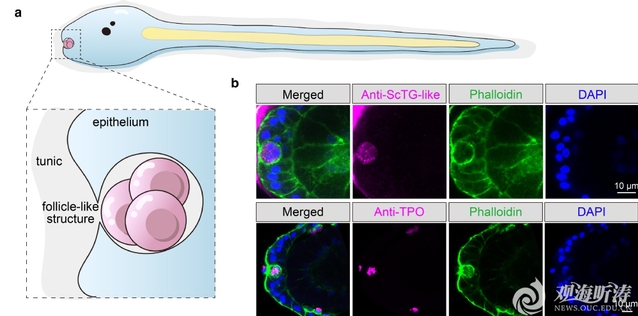
In addition, the team identified an invaginated follicle-like structure in the central anterior region of the larval trunk. This structure consists of three to five closely contacted cells and is highly enriched in THs and proteins key to TH synthesis-related proteins, such as TG-like. Its tissue morphology and molecular composition are highly similar to those of vertebrate thyroid follicles. After knockdown of the Tg-like gene, TH signals in this follicle-like structure became undetectable, and its structural integrity was also severely disrupted. These findings indicate that the follicle-like structure is the major site of TH synthesis and storage in ascidian larvae, representing a proto-thyroid follicle.
At the molecular evolutionary level, based on the domain-based structure of the TG-like protein in S. clava rather than merely primary sequence similarity, the researchers predicted the presence of TG-like proteins with similar structural characteristics across bilaterians. This finding supports the notion that endogenous TH synthesis may have an ancient and conserved evolutionary origin in bilaterians.
Professor Dong Bo and his team have long focused on ascidians as a model for investigating the regulatory mechanisms of metamorphosis. In 2020, the team published the multi-omics data for the highly invasive ascidian Styela clava in Molecular Ecology Resources, revealing the crucial regulatory role of TH signaling in its metamorphosis. In 2024, the team reported the spatial single-cell transcriptomic atlas for the endostyle of S. clava in Science Advances, uncovering the presence of thyroid-homologous cells in the endostyle and their differentiation trajectory. Building on this foundation, the present study is the first to systematically identify and functionally validate the protein precursor required for TH synthesis in an invertebrate, thereby filling a critical gap in research on the biosynthesis mechanism of TH in invertebrates,which is of great theoretical significance for understanding the origin, evolution, and functional emergence of the thyroid endocrine system.








