On April 6, Professor Wang Hongyu from the School of Medicine and Pharmacy and the MOE Key Laboratory of Marine Drugs at Ocean University of China (OUC), in collaboration with Associate Professor Liu Jianbiao from Shandong Normal University, made new progress in the construction of structurally diverse alkanes through ring-opening difuctionalization of methylenecyclobutanes. The related research was published in Nature Communications under the title “Nickel-Catalyzed Ring-Opening Difunctionalization of Methylenecyclobutanes via Strain-Release-Enabled C-C Activation”.
The efficient assembly of molecular complexity from simple building blocks is a significant goal in catalysis, with profound implications for synthetic chemistry, drug discovery, and materials science. In this context, catalytic multicomponent reactions that form multiple carbon–carbon bonds in a single step under mild conditions are desirable for generating molecular complexity because they enhance atom economy, reduce waste, and streamline synthetic design. A breakthrough in this field has been the 1,2-difunctionalization of terminal alkenes and alkynes, whose ready availability has enabled significant advances through transition-metal catalysis, photocatalysis, electrocatalysis, and synergistic techniques. However, these approaches typically produce either two C(sp³)– or two C(sp²)– hybridized carbon centers, thereby limiting access to molecules with differently hybridized carbon groups. The ability to simultaneously build both C(sp³) and C(sp²) centers in one step remains a key challenge. In this regard, terminal allenes stand out as especially useful substrates, offering a solution to this challenge due to their orthogonal π-systems and distinct electronic properties. Their regio- and chemoselective difunctionalization enables the formation of C(sp³)/C(sp²) centers, providing a more straightforward route to densely functionalized, three-dimensional structures.
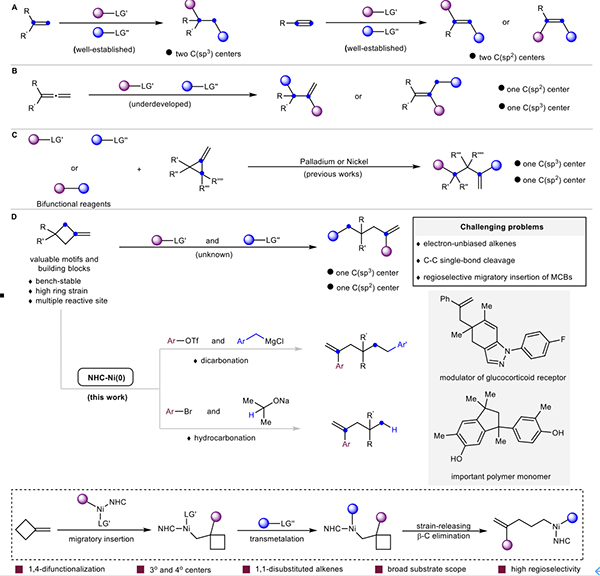
Methylenecyclobutanes (MCBs), as versatile organic reagents, hold considerable potential in chemical synthesis. They offer built-in reservoir of a strain energy (strain energy of cyclobutane: 26.5 kcal/mol, cyclopropane: 27.5 kcal/mol) and dual reactivity through their C=C π-system and cyclobutane σ-system. This duality enables transformations via C=C bond difunctionalization or strain-driven C–C bond cleavage, thus significantly enriching the chemical space of MCBs. To date, extensive studies on the applications of MCBs have been reported. Nevertheless, progress has predominantly focused on 1,2-difunctionalization across the C=C bond and ring-expansion transformations, whereas ring-opening strategies that enable 1,4-difunctionalization of cyclobutanes through selective C–C bond cleavage remain largely underexplored, thereby limiting their utility in multicomponent reactions for molecular complexity.
Given the high ring strain and electronically unbiased nature of the C=C bonds in MCBs, achieving regioselective migratory insertion across these alkenes is both a fundamental challenge and a critical prerequisite for efficient ring-opening functionalization. To address this issue, the researchers developed a Ni–NHC catalysis system that overcomes inherent steric and electronic biases in MCBs. Through branch-selective insertion into the exocyclic methylene group, the system induces strain-driven β-carbon elimination, allowing for direct C–C bond cleavage and subsequent 1,4-difunctionalization. This process produces nonadjacent sp2 and one sp3 carbon centers, which are important building blocks for furnishing many value-added molecules.








