On January 28, a team led by Professor Meng Xiangchao from the College of Chemistry and Chemical Engineering and the MOE Key Laboratory of Marine Chemistry Theory and Technology at Ocean University of China (OUC) published a research article entitled “Modulation of Local Hydrogen Bonding for Highly Efficient Bi-MOFs Photocatalyzed Fixation of N₂ in Aqueous Solution” in the internationally renowned journal Angewandte Chemie International Edition.
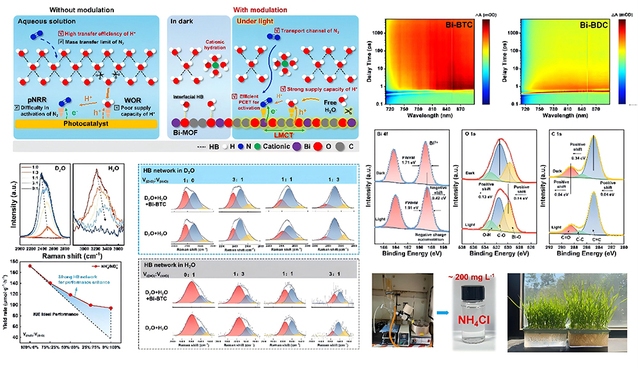
Ammonia is not only an important feedstock for artificial fertilizer and other chemical production in industry, but also a promising hydrogen-fuel carrier. Due to the high energy consumption and massive CO2 emissions in the conventional ammonia synthesis, a green transition is urgently needed. Photocatalytic nitrogen reduction reaction (pNRR), as a potential pathway for converting N2 into ammonia under mild conditions driven by solar energy, is regarded as one of the ideal routes for green ammonia synthesis. In aqueous photocatalytic systems, H2O serves not only as a reactant but also as a solvent. The hydrogen bond (HB) network at the interface between the catalyst surface and local water molecules plays a crucial role in key processes such as proton supply, nitrogen adsorption and activation, and reactant mass transfer. However, balancing nitrogen activation efficiency, proton supply capacity, and proton transfer rate is a central challenge in the field of photocatalytic nitrogen fixation.
To address these challenges, building on their previous work, the team proposed a new strategy in this study to synergistically optimize the HB network and the ligand-to-metal charge transfer (LMCT) effect by regulating the local microenvironment of metal-organic frameworks (MOFs). Using Bi-MOFs as the substrate, the researchers selected the ligand benzenetricarboxylic acid (H3BTC), which exhibits stronger π-electron delocalization, thereby strengthening the LMCT process and promoting the separation and migration of photogenerated carriers. Combined with the unique electronic properties of Bi, this approach significantly promoted N2 adsorption and activation. The study found that, in a weakly alkaline environment, the system not only maintained a high H+ transfer rate but also formed efficient transport channels through cationic hydration effects, thereby facilitating the migration of N2 to active sites. In situ Raman spectroscopy and theoretical calculations revealed that the HB network on the catalyst surface underwent dynamic evolution under illumination, generating free H2O that could participate in the water oxidation reaction (WOR) as reactants, significantly enhancing H+ supply capacity. This process, combined with the LMCT mechanism, effectively facilitated the proton-coupled electron transfer (PCET) process, ultimately achieving a dynamic balance among N2 activation efficiency, H+ supply capacity, and H+ transfer rate. This orchestrated interplay enabled an ammonia synthesis rate of 258.86 µmol·g−1·h−1 at ambient conditions. Furthermore, the researchers used the post-reaction solution for the hydroponic cultivation of wheat seedlings. Compared to the control group cultivated with distilled water, the wheat seedlings grown in the post-reaction solution exhibit clear advantages in germination, root development, and shoot and leaf elongation, demonstrating promising application potential. In addition, rotary evaporation enabled the enrichment of ammonium salts in the post-reaction solution to a concentration of approximately 200 mg·L−1, providing a feasible route for further utilization of the product.







