On February 25, a research team led by Professor Long Hongan from the Institute of Evolution and Marine Biodiversity and the College of Fisheries at Ocean University of China (OUC), together with 14 research institutions in China and abroad, published an article entitled “Cross-domain transfer of trehalose biosynthesis genes contributes to adaptation in high-altitude environments” in National Science Review. In this study, researchers successfully isolated and established a stable culture of the ciliate protozoan Apourosomoida sp.LHA081A01 from Yibug Caka, a Tibetan salt lake at an altitude of 4,533 meters, and systematically elucidated the molecular and evolutionary mechanisms underlying its resilience to multiple extreme stresses, including high salinity, low temperature, and hypoxia.
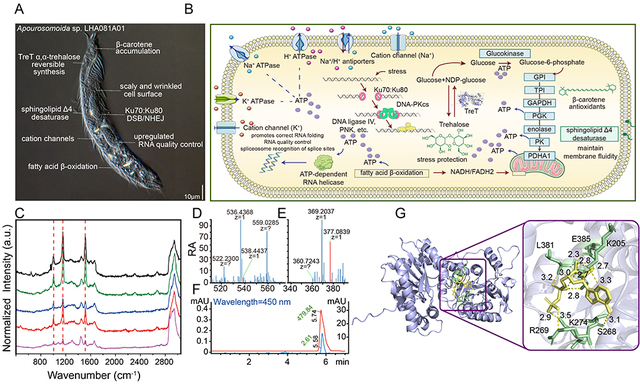
The high salinity, hypoxia, low temperature, and intense radiation of the salt lakes on the Qinghai-Tibet Plateau pose extreme challenges to survival, while also fostering polyextremophiles unique to that region. Owing to difficulties in sampling and technical limitations, the survival strategies of eukaryotic microorganisms in plateau salt lakes, such as protozoa, have long been unstudied.
To uncover the mechanism underlying this stress resilience, the researchers analyzed the ciliate’s unusual nanochromosomes genome, in which each chromosome carries only 1 – 2 genes, and identified a key stress-resistance factor: the TreT glycosyltransferase. Evolutionary analyses showed that about 62.15 million years ago, their ancestors acquired this gene through horizontal gene transfer from a strictly anaerobic and halophilic Desulfobacteraceae bacterium. TreT catalyzes the synthesis of α,α-trehalose—a universal protein stabilizer absent in most other ciliates but essential for counteracting multiple environmental stressors.
Further analyses revealed that this exogenous gene expanded into three copies in the host genome and underwent significant functional divergence: TreT 1709.1 was assimilated into the core metabolic network and contributes broadly to stress tolerance; TreT 6901.1 underwent functional specialization of hypoxia response; whereas the most ancestral-like copy, TreT 9057.1, displayed little apparent contribution to stress tolerance. In addition, this ciliate can remove harmful free radicals by accumulating large amounts of β-carotene and has developed a multilayered tolerance system through multiple membrane-regulatory and gene quality-control mechanisms.

This study not only places high-altitude microbial eukaryotes at the forefront of extremophile research but also provides important insights into the molecular strategies underlying adaptation in extreme life forms. The researchers prospectively pointed out that, in the context of global warming, accelerated glacier melting on the Qinghai-Tibet Plateau may lead to desalination in salt lakes and a regional increase in temperatures, posing severe challenges to this protozoan. Although it possesses multiple stress-resilience mechanisms, it cannot survive for long under low-salinity or freshwater conditions. This finding provides a vivid example of how climate change may reshape the fate of high-altitude microbial eukaryotes.







