Recently, a research team led by Professor Huang Minghua from the School of Materials Science and Engineering at Ocean University of China (OUC) made significant progress in the field of anion exchange membrane water electrolyzers (AEMWEs). The related findings were published in the international journals Angewandte Chemie International Edition and ACS Nano, respectively.
Developing clean and efficient energy-conversion technologies, such as water electrolysis for hydrogen production, is an important pathway toward building a green hydrogen economy. Owing to their low cost and environmental friendliness, AEMWEs have shown broad application prospects for sustainable hydrogen production. However, the cathodic hydrogen evolution reaction (HER) in AEMWEs under alkaline conditions is kinetically hindered by the extra water dissociation step and the complex conversion of multiple reaction intermediates, especially at high current densities. Conventional strategies that optimize only a single step have proved insufficient to overcome this bottleneck. Therefore, it is essential to develop highly efficient HER catalysts featuring cooperative active sites to systematically elucidate the intrinsic coupling mechanisms among multiple HER elementary steps, thereby advancing the development of high-performance AEMWE systems.
To address these challenges, the research team drew inspiration from the concepts of “positive feedback” and “cascade catalysis” in enzymatic systems and introduced them into the area of electrocatalytic HER. They successfully designed a catalyst comprising NiRu dual single-atoms and adjacent NiRu nanoclusters anchored on nitrogen-doped carbon (Ni1Ru1-NiRu@NC). In this catalyst, the atomic Ni and alloyed Ni sites could precisely tailor the electronic structure of neighboring Ru species, giving rise to the corresponding electron-deficient and electron-rich Ru centers that function as Lewis acid and base sites, respectively. The synergistic cooperation between these acid-base sites enables highly efficient hydrogen evolution. Specifically, the Lewis acid Ru sites facilitate the highly effective dissociation of water and continuously supply protons, while the generated protons are swiftly transported through a high-speed hydrogen spillover channel to adjacent Lewis base Ru sites, where they are consumed for hydrogen production. In turn, the rapid proton consumption at the base sites drives further continuous water dissociation at Lewis acid sites, thereby establishing a self-reinforcing catalytic cycle. This catalyst delivers a current density of 1.0 A cm−2 at an overpotential of only 246 mV and exhibits operational stability for over 800 h. More importantly, the AEMWE device assembled with a cathode of Ni1Ru1-NiRu@NC exhibits exceptional long-term stability at 1.0 A cm−2 over 1000 h with a low degradation rate of 0.015 mV h−1. This work proposes a positive feedback-driven coupling strategy that enables cascade acceleration of HER elementary steps, providing a new pathway for the design of highly efficient alkaline HER catalysts and the practical application of AEMWEs. The related results were published in Angewandte Chemie International Edition under the title “Positive Feedback-Driven NiRu Frustrated Lewis Pairs Catalyst Enables a Self-Reinforcing Catalytic Cycle for Cascade-Coupled Hydrogen Production”.
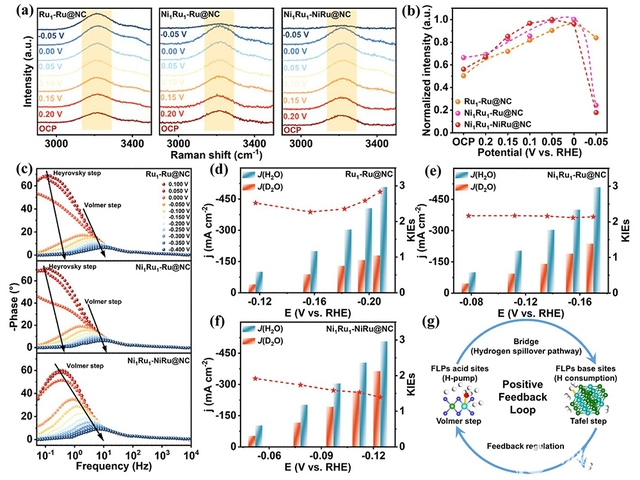
At the same time, the team also developed a frustrated Lewis pairs (FLPs) catalyst based on an amino-functionalized NiRu-based metal–organic framework (MOF). In this catalyst, amino groups and Ni sites spontaneously formed FLPs cooperative active sites, which significantly promote water adsorption and dissociation, generating a locally acidic microenvironment conducive to proton transport, and facilitate continuous proton supply. Meanwhile, the amino groups modulate the electronic structure of neighboring Ru sites, lowering the energy barrier for hydrogen desorption and promoting spillover-mediated hydrogen release, thereby improving the reaction kinetics of HER in alkaline media. Benefiting from these synergistic effects, this catalyst achieves Pt-beyond HER activity and durability under alkaline and neutral conditions at industrial current densities. The assembled AEMWE further demonstrates a low cell voltage (1.76 V at 0.5 A cm–2) and high energy efficiency (71.2%). This study provides new insights into the rational design of efficient and durable HER catalysts. The related results were published in ACS Nano under the title “Frustrated Lewis Pairs Enable Continuous Short-Range Hydrogen Spillover for Industrial-Scale Hydrogen Evolution”.
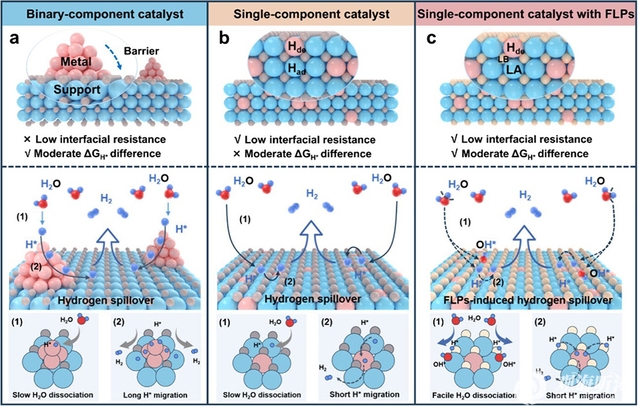
These findings deepen our understanding of the cooperative catalytic mechanisms and structure-performance relationships of frustrated Lewis pairs catalysts in efficient hydrogen evolution, providing key theoretical support and material foundations for the practical development of anion exchange membrane water electrolysis for hydrogen production.







