The emergence of biomineralized skeletons was one of the hallmark events of the Cambrian Explosion and profoundly shaped the basic pattern of animal biodiversity. Such structures evolved independently multiple times across different animal phyla and display convergent evolution, suggesting that a biomineralization regulatory “toolkit” may have already existed in the common ancestors of animals and was repeatedly co-opted to provide the genetic and regulatory foundation for the subsequent evolution of skeletal structures. However, this hypothesis has long lacked systematic molecular evidence from the perspective of gene regulation.
On January 20, a research team led by Professor Li Qi from the MOE Key Laboratory of Mariculture at Ocean University of China (OUC) published an article in Molecular Biology and Evolution under the title “Evolutionary innovation within conserved gene regulatory networks underlying biomineralized skeletons in Bilateria”. Using the oyster Crassostrea nippona as a model, the study systematically characterized the chromatin landscape and gene regulatory networks (GRNs) underlying primary larval shell and secondary adult shell formation, and proposed an evolutionary model in which conserved GRNs form the core framework while dynamic chromatin remodeling drives phenotypic innovation.
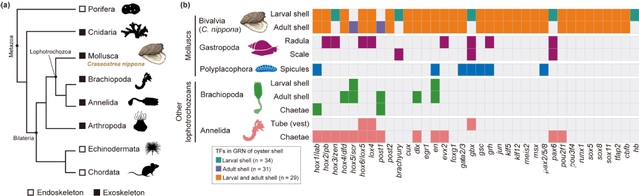
The study found that although larval and adult shells differ markedly in morphology, structure, and protein composition, the upstream transcription factor networks underlying their formation are highly conserved and originated before the emergence of molluscs. These transcription factors (TFs) show relatively stable expression across developmental stages, whereas the effector genes directly involved in biomineralization exhibit pronounced stage-specific expression. Further analyses showed that stage-specific remodeling of regulatory element activity enables dynamic chromatin landscapes to reconnect conserved TFs with rapidly evolving effector genes, thereby switching biomineralization programs across life stages and generating diverse skeletal architectures from the same underlying genetic framework. Cross-species comparisons further showed that these key TFs are also widely recruited during larval exoskeleton formation in other lophotrochozoans, supporting the existence of a conserved biomineralization regulatory “toolkit” in lophotrochozoans. By further comparing the GFNs underlying biomineralized skeleton formation in molluscs and echinoderms, the researchers found that although the two groups differ fundamentally in structure and developmental trajectory, both achieve convergent evolution of skeletal traits through heterochronic deployment of ancestral TFs. This suggests that upstream regulatory modules are under deep evolutionary constraint, whereas phenotypic diversity is primarily driven by the remodeling of downstream effector genes and the regulatory elements.
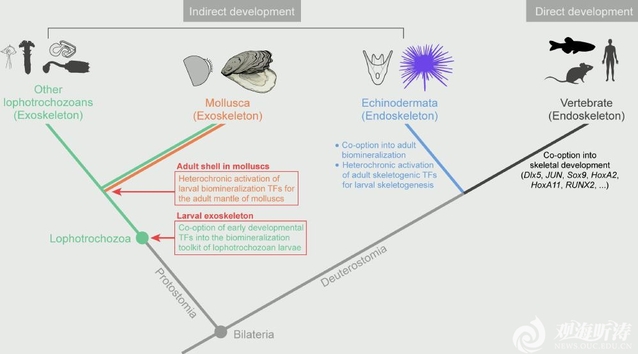
This study reveals the key role of dynamic chromatin states in driving modular innovation of biomineralized skeletons within a conserved genetic framework. It deepens our understanding of the relationship between evolutionary constraint and genetic innovation, and provides important clues for understanding the evolutionary mechanisms underlying complex animal traits.







