On October 4, 2025, a research team led by Professor Han Lu from the School of Medicine and Pharmacy, the MOE Key Laboratory of Marine Drugs, and the Qingdao Marine Science and Technology Center at Ocean University of China (OUC) developed a reactive oxygen species (ROS) self-driven nanomotor that operates in the pathological microenvironment generated by thrombosis and, in combination with near-infrared (NIR) irradiation, enables synchronized thrombolysis and vascular remodeling. The findings were published in Advanced Materials in an article entitled “NIR-Driven Nanomotors Integrating With Platelet-Thylakoid Hybrid Membranes for Synchronized Thrombolysis and Vascular Remodeling”. Nanowerk reported and commented on the study in its Spotlight column in an article entitled “Light-driven nanomotors clear blood clots and restore vessel health”.
The incidence of thrombotic cardiovascular diseases is increasing, seriously influencing human health. Vascular endothelial injury induced by inflammation, oxidative stress, hypertension, hyperglycemia, hypoxia, and other factors might result in vascular obstruction, blood flow disruption, and ischemic organ damage. Critically, injured endothelial cells at the thrombus site can trigger inflammatory damage and generate high concentrations of hydrogen peroxide (H2O2). This self-perpetuating cycle of oxidative stress exacerbates endothelial dysfunction through inflammatory cytokine release and platelet activation. Currently, most antithrombotic strategies focus predominantly on clot dissolution, while leaving the underlying pathological microenvironment unresolved. Consequently, endothelial injury often persists, predisposing patients to thrombus recurrence. Thus, the development of nanotherapeutics capable of simultaneously dissolving clots and repairing the vascular endothelium represents an urgent clinical need.
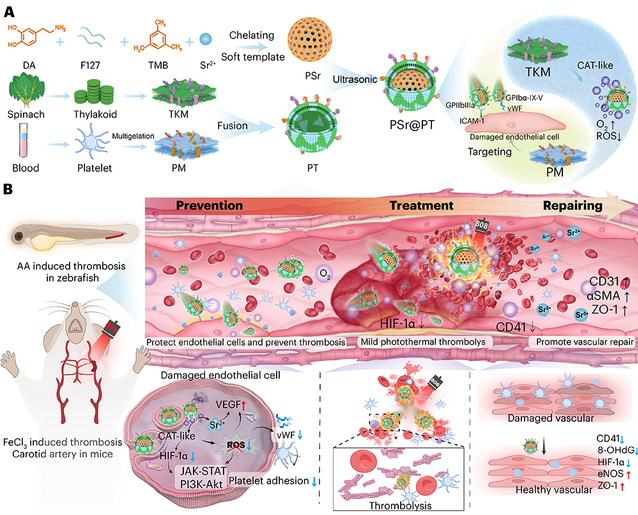
In this study, the team successfully engineered a pathogenesis-driven nanomotor system (PSr@PT NPs). Its core is composed of strontium (Sr)-doped mesoporous polydopamine (PSr NPs), which acts both as a nanomotor base and as a reservoir for oxidative stress-triggered Sr2+ release, while simultaneously serving as a ROS scavenger. To achieve dual-targeted propulsion, a hybrid cloak integrating platelet membranes (PM) and spinach-derived thylakoid membranes (TKM) was constructed on the surface of PSr NPs. PM enables precise homing to activated endothelial cells at thrombotic sites, whereas TKM confers catalase-like activity, continuously decomposing H2O2 into O2 to power autonomous motion. Ultimately, the system mitigates platelet-endothelial adhesion by suppressing inflammation and oxidative stress. Upon NIR irradiation, the PSr core generates mild photothermal heating (approximately 40 °C), achieving dual thrombolytic effects through direct thermal clot dissolution and improved nanoparticle infiltration. Concurrently, controlled Sr2+ release from the PSr NPs promotes endothelial regeneration and barrier restoration, thereby interrupting the cycle of thrombosis recurrence. The universal thrombolytic efficacy of this nanosystem was validated across species-specific models, including arachidonic acid (AA)-induced thrombosis in zebrafish and ferric chloride (FeCl3)-induced carotid embolism in mice. By combining the catalytic functions of plant photosynthetic membranes with the targeting precision of mammalian systems, this cross-species synergistic strategy establishes a new paradigm in nanobionics. This biomimetic structure organically integrates biological signal transduction (platelet-endothelial recognition) with physical stimulus responsiveness (photothermal driving), reshaping the thrombotic microenvironment while promoting deep penetration into clots. This multidimensional treatment strategy upgrades “instant thrombolysis” to “sustained vascular functional recovery”, providing a nanoplatform with translational potential for precise antithrombotic therapy.