On March 20, a research team led by Taishan Scholar Chen Xiguang and Professor Liu Ya from the College of Marine Life Sciences at Ocean University of China (OUC) published their latest findings in Biomaterials under the title “Reassembly nanomaterials-mediated engineered bacteria lysis for reshaping immunosuppressive microenvironment”.
One of the major challenges facing tumor immunotherapy is the immunosuppressive microenvironment of “cold” tumors, which is characterized by a lack of immune cell infiltration and insufficient inflammatory signals. Bacterial therapy, owing to the inherent tumor-homing and immunomodulatory capabilities, has attracted considerable attention. However, conventional engineered bacteria are still limited by interference from the tumor microenvironment that affects gene expression, the potential tumor-promoting risks of bacterial metabolites, and the biosafety concerns associated with live bacteria. Therefore, developing a novel bacteria-nanomaterial hybrid system capable of controllable drug release, enhanced immune activation, and improved safety has become key to overcoming these bottlenecks.
In this study, the researchers developed a bacteria-nanomaterial hybrid system, IE-PPCs. By transforming the IFN-γ gene into E. coli Nissle 1917 (EcN), the researchers obtained the engineered bacterium IE and anchored it onto the polymer-peptide conjugates (PPCs) with matrix metalloproteinases (MMPs)-responsive bacteria through amide bonds. Upon intravenous injection, the IE-PPCs system is selectively colonized into the tumor microenvironment, where MMPs are highly expressed. The PPCs spontaneously transformed into fibrous nanostructures, exposing antimicrobial peptide residues that specifically lyse the bacteria, thereby releasing IFN-γ and bacterial lysates. Both in vitro and in vivo experiments showed that IE-PPC2 systems successfully achieved MMP-responsive bacterial lysis and triggered IFN-γ release. The bacterial lysates and released IFN-γ synergistically polarized M1-type TAM, induced dendritic cells (DC) maturation, shifting the tumor microenvironment (TME) from “cold” into a more immunologically active “hot” state. In 4T1 mouse breast cancer models, IE-PPC combined with anti-PD-L1 therapy achieved 89.7% anti-tumor rate, and bacterial loads within tumors decreased by 98.9%. The treatment also induced long-term immune memory, effectively suppressed tumor recurrence and metastasis, and showed good biosafety.
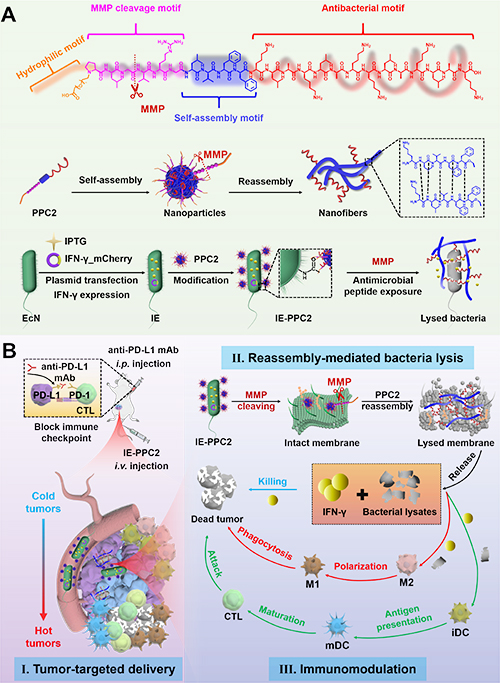
The schematic illustration shows the design and working mechanisms of the IE-PPCs system. In Figure A, PPCs, which were conjugated by antimicrobial peptides, enzyme-cleavable peptides, self-assembly motif (LVFF), and hydrophilic PEG, self-assembled into nanoparticles in solution and were then covalently attached to the surface of IPTG-induced IE expressing IFN-γ via amide bonds, thereby forming IE-PPC2. Figure B illustrates its triple-cascade mechanism in vivo. First, after intravenous injection, IE-PPC2 actively targeted the tumor site. Then, in the MMP-rich tumor microenvironment, PPC2 underwent reassembly from nanoparticles into nanofibers, exposing the antimicrobial peptides and specifically inducing IE lysis, thereby releasing IFN-γ and bacterial lysates. Finally, these released components act synergistically to promote DC maturation and M1-type macrophage polarization, enhance antigen presentation, and, in combination with anti-PD-L1 antibody-mediated immune checkpoint blockade, remodel the immunosuppressive “cold” tumor into T-cell-infiltrated “hot” tumors, thus enabling efficient and safe antitumor immunotherapy.
Research Highlights and Innovations
First, an intelligent spatiotemporally controlled lysis mechanism. One of the key innovations of this study is the design of the MMP-responsive reassembly nanomaterial PPC2. Distinct from conventional synchronous lysis circuits or external physical triggers, the system exploits highly expressed MMPs in TME as an endogenous “switch”. Upon MMP stimulation, PPC2 transforms in situ from nanoparticles into nanofibers, exposing antimicrobial peptides and thereby triggering bacterial lysis. This design enables precise spatiotemporal control over bacterial lysis and therapeutic release, avoids the nonspecific lysis of bacteria in off-target tissues, and significantly enhances the therapeutic specificity and safety.
Second, a synergistic immune-microenvironment remodeling strategy. Following bacterial lysis, the system enables the synchronized and localized release of IFN-γ and bacterial lysates, including PAMPs such as lipopolysaccharides (LPS) and DNA. This combination does not simply add together separate functions; rather, it generates a strong synergistic effect. IFN-γ directly inhibits tumor cell proliferation and promotes M1 macrophage polarization, while bacterial PAMPs serve as potent natural immune adjuvants that promote the DC maturation and antigen presentation. Through their combined action, the system effectively reverses the immunosuppressive tumor microenvironment, drives the polarization of M2 macrophages toward the M1 phenotype, and significantly increases CD8+ T cell infiltration, thereby achieving full-chain immune activation from innate immunity to adaptive immunity.
Third, a design that integrates safety control with long-term protective effects. The team prospectively addressed the central challenge of live bacterial therapy: safety. By integrating the therapeutic function with the bacterial lysis program, IE-PPC2 can be effectively cleared by the immune system after completing its therapeutic task. Experimental data showed that intratumoral bacterial loads decreased by 98.9% after treatment, while bacterial residues in major organs remained minimal, effectively reducing the risks of bacteremia and excessive inflammatory responses. More importantly, the robust immune response induced during pathogen clearance successfully established long-term immune memory, as evidenced by significant increases in effector memory T cells and central memory T cells. This conferred strong protection against tumor recurrence and distant metastasis, reflecting an advanced design concept that balances both treatment efficacy and long-term protection.







